contact
0769-83699986
135-3754-4550(mr. zhu)
at present, there are many safety standards for lithium batteries worldwide, but in reality, power batteries that meet safety standards cannot guarantee their safety even after strict testing and verification.
is the battery safety standard effective
in fact, safety testing cannot guarantee the safety of power batteries in practical applications. this is because the safety of lithium-ion batteries is divided into abuse safety and on-site safety. the so-called abuse of safety refers to mechanical problems such as compression, needle puncture, short circuit, overcharging, overheating, hot box, fire, etc; on site safety is caused by manufacturing defects, such as connection issues, diaphragm damage, dust, etc. these problems occur randomly, causing internal short circuits and resulting in overheating and thermal runaway.
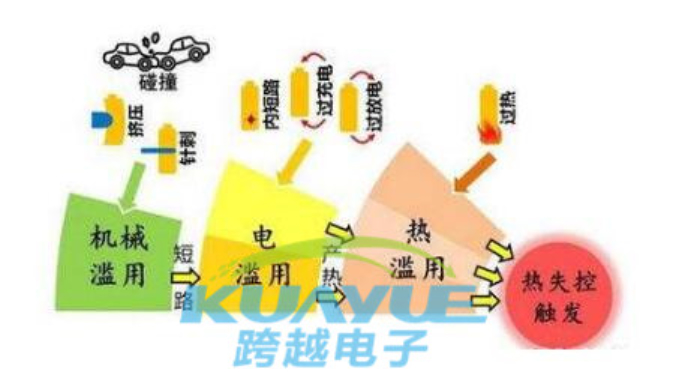
from a safety perspective, the abuse of security is predictable, and each battery can be evaluated through testing. the occurrence process is long and can be improved through protective measures; however, self triggering safety is unpredictable, occurring randomly with a small probability, and cannot be evaluated through testing or completely eliminated through quality management. at present, all safety measures cannot completely eliminate the safety hazards of power batteries.
taking the safety reality of lithium-ion batteries as an example, lithium-ion batteries that have experienced safety accidents have all passed safety certification before. for laptop batteries, the probability of occurrence is one in a few million; if we simply magnify the probability of accidents based on battery capacity, the statistical probability of single cell accidents for 18650 batteries is about one in ten million. by simply adding up the actual number of 18650 batteries in the battery pack, the probability of battery accidents can be roughly calculated. however, in practical applications, it is definitely much higher than the calculated value. the cause of such accidents is basically unpredictable internal short circuits, which cannot be completely eliminated. the batteries that cause safety accidents are all qualified products during manufacturing.
in the field of power batteries, the heat dissipation of large batteries (groups) is much more difficult than that of small batteries (individual); the power battery management system is more complex, and its effectiveness and reliability are reduced; the usage environment of power batteries is more harsh (high and low temperature, vibration, collision); the power battery requires a long service life, and the problem is more severe in the mid to late stages of its lifecycle.
overall, there is no "scientific" connection between the test results of abusing security standards and the occurrence or non occurrence of security incidents.
mechanism of thermal runaway caused by internal short circuit
the safety on short circuits seems to be caused by multiple factors working together so far. there are many reasons for defects in the manufacturing process, application process, design process, and some improper applications. unlike external short circuits with uniform heating, internal short circuits are high temperatures at local points.
the risk assessment of internal short circuit mainly includes: low-temperature lithium deposition, negative electrode metal deposition, charging lithium deposition, data weighting, micro short circuit, and thermal stability. the most important issue is whether lithium will precipitate during low-temperature charging, even tiny metal pollutants that cannot be detected by the naked eye can cause internal short circuits.
positive electrode metal pollutants - chemical internal short circuits: even tiny metal pollutants such as iron, chromium, nickel, copper, zinc, etc. that cannot be detected by the naked eye can cause internal short circuits. during battery charging, pollutants are oxidized at the positive electrode and become ions that enter the solution. under the action of an electric field, they move to the negative electrode and obtain electrons on the surface of the negative electrode, which are reduced to metal. they continue to grow, pierce the separator, and form short circuits, manifested as severe self discharge or thermal runaway.
there are many possible ways for metal foreign objects to enter lithium electronics, and automotive batteries can be transmitted to battery manufacturing through air at any time during production or operation. therefore, in order to do a good job in batteries, the quality control of the production environment needs to be very strict.
the most intuitive way to judge whether a battery is good or not is to open and inspect the separator. if there are no stains, then such a battery is much better than one with many stains after opening. this is because the positive and negative electrodes of the battery have a deposition effect during the charging and discharging process. after a certain degree of cycling of the deposition effect, it will cause a short circuit and the battery will explode. many battery accidents, he xiangming believes, may be caused by this factor, so when designing batteries, this factor must be considered.
control and avoid thermal runaway pathways of batteries
at present, determining some key parameters of the thermal runaway process and establishing a "scientific" connection with the evolution process of thermal runaway are the main means to solve the thermal runaway of power batteries.
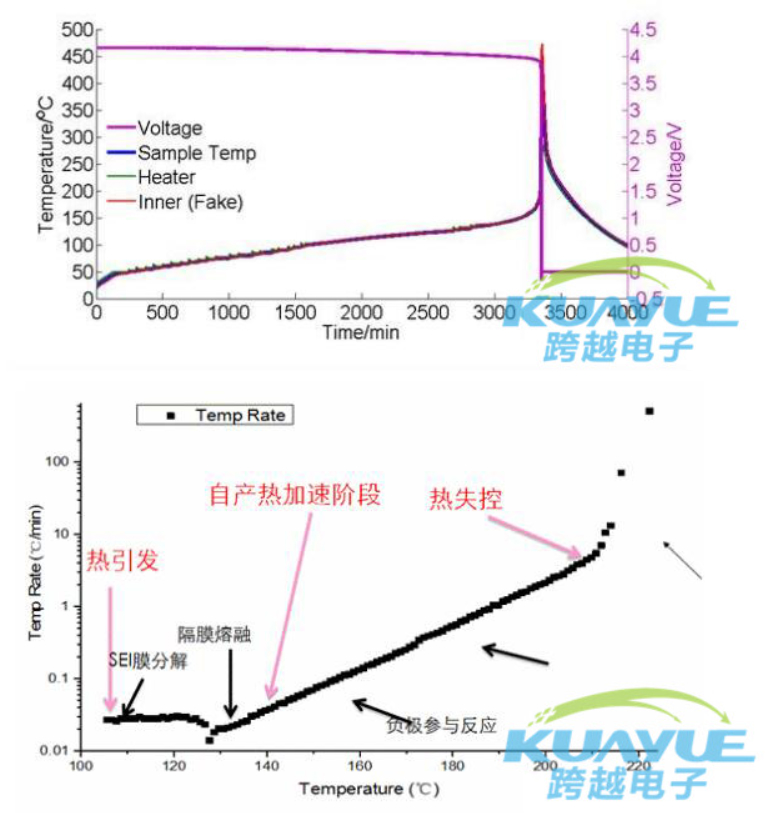
in addition, from the actual usage status of power batteries, many people mistakenly believe that the higher the energy of the battery, the less safe it is, and this statement has no scientific basis. in fact, the safety of battery thermal stability is related to the production enterprise, not the material.
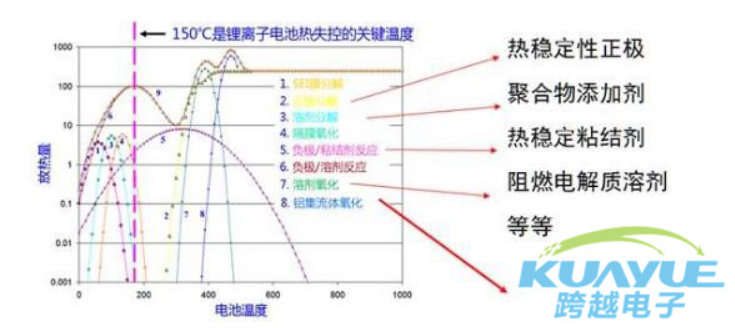
this is because the fundamental reason for thermal runaway is that the process of battery thermal runaway is a "chain" exothermic reaction. the solution is to reduce the exothermic reaction and cut off the "chain" exothermic reaction. the coping strategies include reducing the heat released from internal chemical reactions, decreasing the heat released from internal chemical reactions, increasing the temperature at which chemical reactions occur, reducing the rate of battery temperature rise, and enhancing external heat dissipation of the battery. by taking these measures and changing the path of the chain, the battery can be made safer.
contact
0769-83699986
135-3754-4550(mr. zhu)

business

business 1

business 2

business 3

technology